
To maximize the total spin, the electrons in orbitals with only one electron all have the same spin (or the same values of the spin quantum number).It states that before a second electron is filled in an orbital, every orbital in a given subshell is singly occupied by electrons.Hund’s rule specifies the order in which electrons are filled in all subshell orbitals.
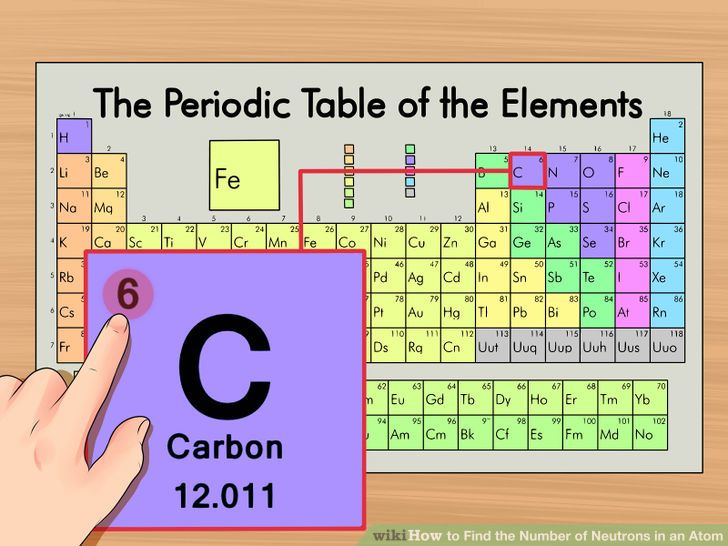

Subshell labels are used to write down an atom's electron configuration.As a result, the 1p, 2d, and 3f orbitals do not exist because the value of the azimuthal quantum number is always less than the value of the principal quantum number.
